
This video follows up on two videos introducing molecular and electron geometry. Because we're saying try that would mean that the electronic geometry is tribunal planer. In this video we learn how to use electron and molecular geometry charts. Now, if we take a look, we'd say that the carbon is connected to how many electron groups It's connected to one, 2. So this would represent the structure or molecular shape of formaldehyde. What has six valence electrons and here we're showing all of them. So carbon would have to form a double bond to that oxygen. It's already making two bonds and eat two more. Fill in the following tables (follows octet rules): of electron. The hydronium ion, H 3 O +, forms when acids are dissolved in water. The chart below depicts some molecule geometries based on the number of electron pairs present. (b) Two of the electron regions are lone pairs, so the molecular structure is bent. The angles between all neighboring groups 90 ° while the ones separated with an atom is 180 o. 9: (a) H 2 O has four regions of electron density around the central atom, so it has a tetrahedral electron-pair geometry. A good example in general chemistry is sulfur hexafluoride, SF 6: Four of the groups lie in a single plane, and the other two are above and below it. Mhm Carbon itself has to make four bonds. Identify: the electronic geometries (i.e., takign into account the electrons) molecular geometries (only the nuclei) relevant bonding angles. Let’s put a little chart of example for steric number ranging from 2-6. Visualize trends, 3D orbitals, isotopes, and mix compounds. And Remember Hydrogen Czar Group one and they can only form single bonds. Interactive periodic table showing names, electrons, and oxidation states. So as a result it's carbon that has to go in the center. But remember we have another rule when it comes to hydrogen, it can never go in the center. Um So if we take a look here, we place the least electro negative element in the center. So for this example, it says determined the electron geometry for the following molecule CH two. Electrons, whether bonded or in lone pairs, will repel each other, and they arrange around a central atom in a way that minimizes this repulsion and maximizes the distance between them. But there are rules to draw the best structure. With fewer 90° LP–BP repulsions, we can predict that the structure with the lone pair of electrons in the equatorial position is more stable than the one with the lone pair in the axial position.Recall there are many possible lewis dot structures that exist. If we place it in the axial position, we have two 90° LP–BP repulsions at 90°. Difference between electron and molecular geometry. 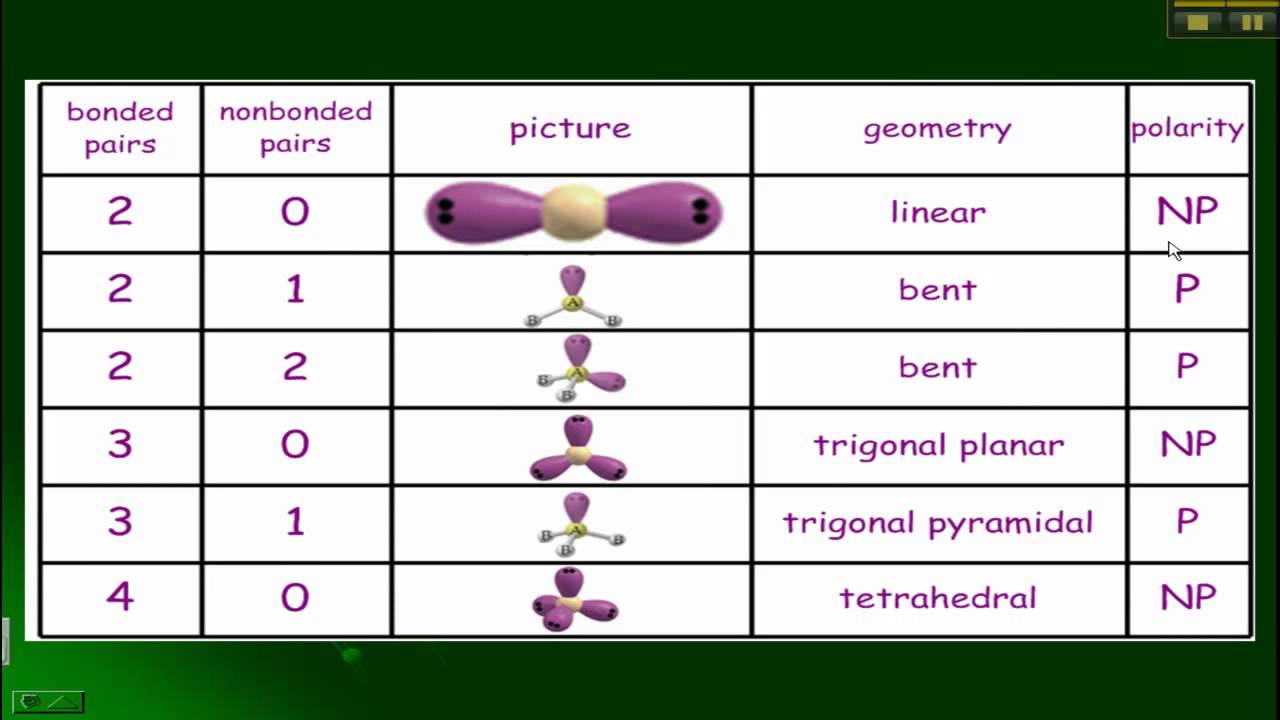
However, because the axial and equatorial positions are not chemically equivalent, where do we place the lone pair? If we place the lone pair in the equatorial position, we have three LP–BP repulsions at 90°. Difference between electron and molecular geometry Electron geometry and hybridization chart Geometry molecular electronic chart electron bonding bond groups angles. The 25+ best molecular geometry ideas on pinterest14. We designate SF 4 as AX 4E it has a total of five electron pairs.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
